|
11/9/2023 0 Comments Solid liquid solid: A substance that retains its size and shape without a container a substance whose molecules cannot move freely except to vibrate.gas: A substance that can only be contained if it is fully surrounded by a container (or held together by gravitational pull) a substance whose molecules have negligible intermolecular interactions and can move freely.It takes the shape of its container but maintains constant volume. liquid: A substance that flows and keeps no definite shape because its molecules are loosely packed and constantly moving.Gaseous matter is composed of particles packed so loosely that it has neither a defined shape nor a defined volume.Particles can move about within a liquid, but they are packed densely enough that volume is maintained. Liquid matter is made of more loosely packed particles.A solid will retain its shape the particles are not free to move around. Solid matter is composed of tightly packed particles.Matter can exist in one of three main states: solid, liquid, or gas.Solid-liquid equilibria in binary mixtures of organic compoundsįluid Phase Equilib., 113, 5, 117 125 (1995)Įxcess Volumes for Alkanol + Morpholine Systems at 298.15 and 308. Solid-Liquid Equilibria for Different Heptanones with Benzene, Cyclohexane, and Ethanol Solid-Liquid Equilibria of Viscous Binary Mixtures with Alcohols 
Solid-Liquid Equilibria of Several Binary Systems with Organic Compounds Solid-Liquid Equilibria for Seven Binary Systemsīinary Solid-Liquid Equilibria of Organic Systems Containing Different Amides and Sulfolane Wittig R., Constantinescu D., Gmehling J.īinary Solid-Liquid Equilibria of N,N-Dimethylacetamide with 1,2-Dichloroethane, Dichloromethane, and 1-PropanolĪtik Z., Ahlers J., Lohmann J., Gmehling J. Teodorescu M., Wilken M., Wittig R., Gmehling J.įluid Phase Equilib., 204, 6, 267 280 (2003)īinary Solid-Liquid equilibria of Organic Systems Containing e-Caprolactone Solid-Liquid Equilibria for Binary Organic Systems Containing 1-Methoxy-2-propanol and 2-Butoxy EthanolĪctivity Coefficient at Infinite Dilution, Azeotropic Data, Excess Enthalpies and Solid-Liquid-Equilibria for Binary Systems of Alkanes and Aromatics with Estersįluid Phase Equilib., 230, 1-2, 131-142 (2005)Īzeotropic and solid - liquid equilibria data for several binary organic systems containing one acetal compound Vapour-liquid equilibria, azeotropic data, excess enthalpies, activity coefficients at infinite dilution and solid-liquid equilibria for binary alcohol-ketone systemsįluid Phase Equilib., 267, 2, 119 126 (2008) 
Machado J.J.B., de Loos T.W., Ihmels E.C., Fischer K., Gmehling J. High pressure solid-solid and solid-liquid transition data for long chain alkanes Solubility and density measurements of palmitic acid in supercritical carbon dioxide + alcohol mixturesīrandt L., Elizalde-Solis O., Galicia-Luna L.A., Gmehling J.įluid Phase Equilib., 289, 1, 72 79 (2010) Selected Scientific Papers (Experimental Data) 2010 Gmehling J.G., Anderson T.F., Prausnitz J.M. 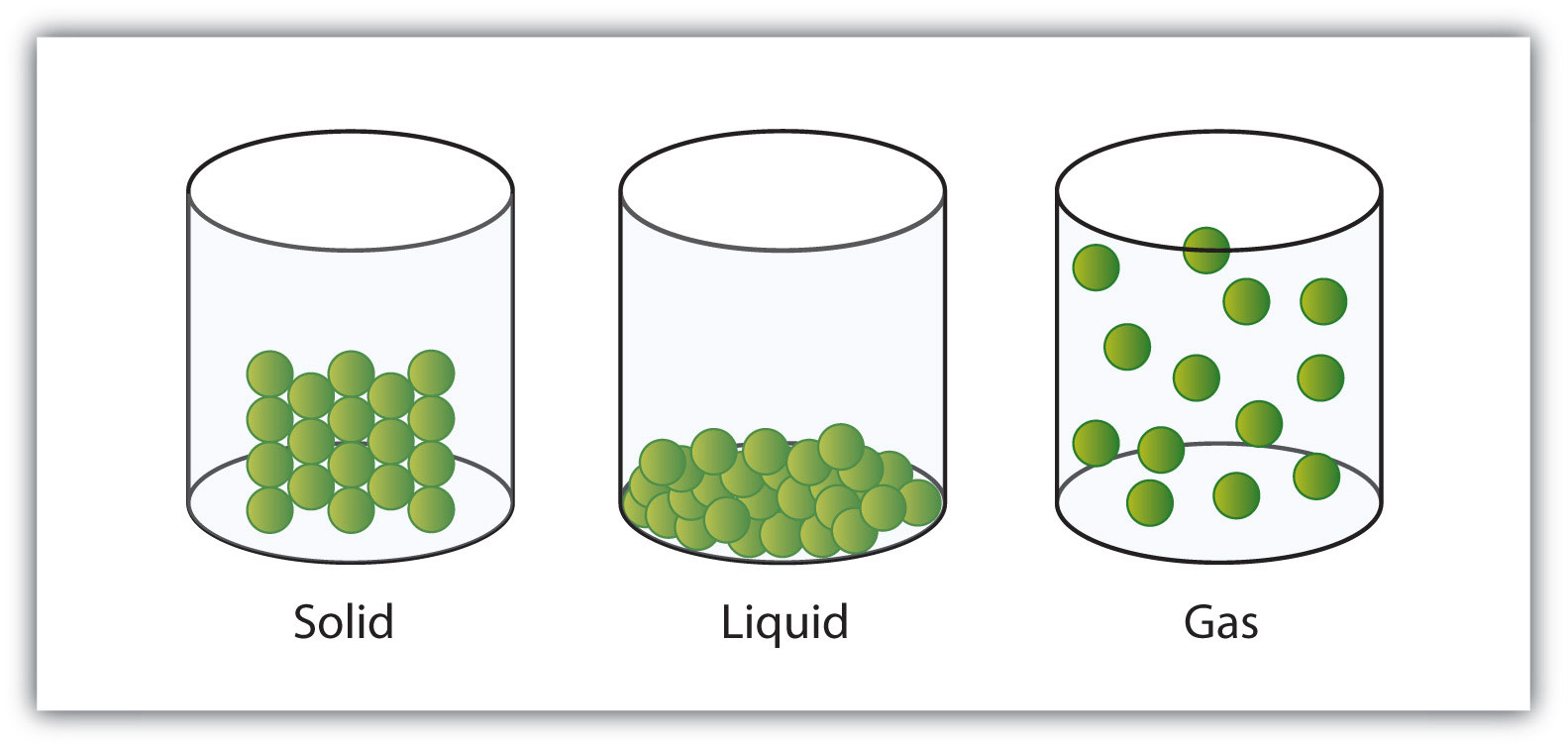
Measurement and Prediction of Ternary Solid-Liquid Equilibria 
The effect of gas pressure on the melting behavior of compoundsįluid Phase Equilib., 210, 2, 199 214 (2003)Įstimation of Enthalpies of Fusion, Melting Temperatures, Enthalpies of Transition, and Transition Temperatures of Pure Compounds from Experimental Binary Solid-Liquid Equilibrium Data of Eutectic Systems Prediction of Salt Solubilities in Aqueous Electrolyte Systems GE Model for Single- and Mixed-Solvent Electrolyte Systems. Prediction of solubilities of salts, osmotic coefficients and vapor-liquid equilibria for single and mixed solvent electrolyte systems using the LIQUAC modelįluid Phase Equilib., 275, 1, 8 20 (2009) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
